URETERAL STENTING KIT / URETERAL DOUBLE PIGTAIL CATHETER
1. DEVICE DESCRIPTION
Ureteral stenting kit consists of a single-channel, radiopaque, polyurethane catheter, clamps, a guidewire, a pusher and a driving ring. The catheter has two pigtail ends with multiple openings located inside the loop, arranged along the axis and spirally along the stem. All sizes of catheters are available with open tips and open-closed tips.
-
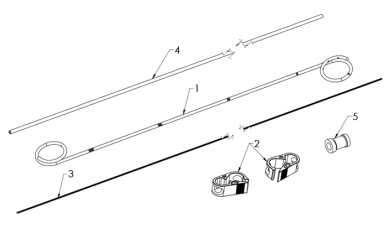
Fig. 1 Elements of the kit
- Catheter
- Clamps
- Guidewire
- Pusher
- Driving ring
-

Fig. 2. Ureteral double pigtail catheter
- Catheter
2. PRODUCT RANGE
All available configurations of this device are presented in Table 1.
Table 1. Available versions
CATHETER SIZE
[F]
DISTANCE BETWEEN LOOPS
[cm]
LOOP DIAMETER
[cm]
3
10-18
2 lub 4
4
16-20
2 lub 4
4.8
20-30
2 lub 4
5
24-30
2 lub 4
6
24-30
2 lub 4
7
24-30
2 lub 4
8
24-30
2 lub 4
8.5
24-30
2 lub 4
10
24-30
2 lub 4
3. INTENDED PURPOSE / INDICATIONS
The device is intended for short-term (up to 30 days) use to ensure the free flow of urine from the kidneys to the bladder in case of obstruction or narrowing of the ureter.
3.1. CONTRAINDICATIONS
The use of the device is usually contraindicated in the following cases:
- bladder fistula;
- bladder outlet obstruction;
- spastic / neurogenic bladder;
- coagulation disorders or the use of anticoagulants;
- allergy or hypersensitivity to polyurethane.
3.2. POTENTIAL ADVERSE EVENTS
Adverse events that may be associated with the use of the device include (in alphabetical order), but are not limited to:
- allergic reaction or hypersensitivity to polyurethane;
- bleeding;
- catheter dislocation;
- catheter fracture;
- catheter incrustation, malfunction and possible retention;
- fever;
- hematuria;
- infection / pyuria;
- pain;
- perforation of the renal pelvis / ureter;
- septic shock;
- symptoms of irritation of the bladder;
- uretero-arterial fistula;
- urinary incontinence / urgency;
- urine extravasation;
- vesico-renal reflux.
Any serious incident that has occurred in relation to the device should be reported to the manufacturer at reklamacje@balton.pl and the competent authority of the country in which the user and/or patient is established.
3.3. INTENDED USER PROFILE
Intended users of this device are only physicians who have received appropriate training in endourological procedures.
3.4. USE ENVIRONMENT
Use of this device is allowed only in healthcare facilities prepared for this kind of urological procedures.
3.5. PATIENT TARGET GROUP
Target group are patients with impaired free flow of urine from the kidneys to the bladder. No known data restricting use of this device in patients of particular gender or race. Before making decision concerning use of this device, potential benefits and risks should be considered individually for every patient.
4. WARNINGS
- This device is designed for single use in a single patient only. Do not resterilize or reuse. Reuse or resterilization may compromise the structural integrity of the device and/or lead to device failure which, in turn, may result in patient injury, illness or death. Reuse or resterilization may also create a risk of contamination of the product and cause patient infection including the transmission of infectious disease(s). Contamination of the product may lead to injury, illness or death of the patient.
- Do not use if the package is opened or damaged.
- Do not use after expiration date given on the package label.
- Do not use if labeling is incomplete or illegible.
6. HOW SUPPLIED
6.1. CONTENT OF THE PACKAGING
One (1) ureteral stenting kit in a blister / one (1) ureteral double pigtail catheter in a pouch, packed with Instructions for Use in a case box.
6.2. STERILITY
This product is supplied sterilized with ethylene oxide gas, in a blister / pouch. Only the content of the blister / pouch should be considered sterile. The device is only sterile if this packaging is not opened or damaged.
7. HANDLING AND STORAGE
Store at room temperature in a dry place, in the case box, as supplied. Do not expose to temperatures outside the range: 10 ºC ÷ 30 ºC.
8. DISPOSAL INSTRUCTIONS
Used product shall be treated as medical waste. After use, dispose the product and packaging in accordance with healthcare facilities, administrative and/or local government policy.
9. WARRANTY
If delivered product is damaged or has any other defects, inform the manufacturer and keep the device with original packaging.
10. OPERATIONAL INSTRUCTIONS
Balton sp. z o.o. shall not be liable for any direct, incidental or consequential damages resulting from the misuse of this product.
- Check the primary packaging (blister / pouch) for possible damage and expiry date.
If there is a suspicion that sterility may be compromised and/or the expiry date has been exceeded it must not be used.
- Open the package and take out the device.
- Check if the device is not damaged.
If there is a suspicion that the device is damaged it must not be used.
- Prepare the component, maintaining sterility.
- Disinfect the perineum according to procedures.
- Cover the patient with sterile drape.
- Remove the guidewire from the protector.
- Put the guidewire into the catheter, until both pigtails are straightened.
- Close the clamp immobilizing the catheter on the guidewire.
- Put the pusher onto the guidewire and advance until it contacts the catheter.
- Close the second clamp immobilizing the pusher on the guidewire.
- Insert the cystoscope into the bladder.
- Insert the prepared kit into the cystoscope, using driving ring.
- Drive the guidewire with the catheter through the ureter, into the renal pelvis.
- Open both clamps.
- Keeping the catheter in the renal pelvis with the use of the pusher, gently withdraw the guidewire and then the pusher.
- Confirm the correct position of the catheter in ultrasound, fluoroscopy or abdominal X-ray.
Regular control of ureteral catheter position, patency and urine retention are indispensable for correct functioning of the ureteral catheter.
SYMBOLS GLOSSARY
-
CE mark
-
Medical device
-
Unique device identifier
-
Manufacturer
-
Date of manufacture
-
Use-by date
-
Catalogue number
-
Batch code
-
Consult instructions for use or consult electronic instructions for use
-
Do not use if the packaging is damaged and consult instructions for use
-
Caution
-
Sterilized using ethylene oxide
-
Do not resterilize
-
Do not re-use
-
Single sterile barrier system
-
Single sterile barrier system with protective packaging outside
-
Non-pyrogenic
-
Keep away from sunlight
-
Fragile, handle with care
-
Temperature limit
-
Not made with natural rubber latex
-
Keep dry
-
Recyclable packaging material
-
n units per package
1. DEVICE DESCRIPTION
Ureteral stenting kit consists of a single-channel, radiopaque, polyurethane catheter, clamps, a guidewire, a pusher and a driving ring. The catheter has two pigtail ends with multiple openings located inside the loop, arranged along the axis and spirally along the stem. All sizes of catheters are available with open tips and open-closed tips.
-

Fig. 1 Elements of the kit
- Catheter
- Clamps
- Guidewire
- Pusher
- Driving ring
-

Fig. 2. Ureteral double pigtail catheter
- Catheter
2. PRODUCT RANGE
All available configurations of this device are presented in Table 1.
Table 1. Available versions
| CATHETER SIZE [F] |
DISTANCE BETWEEN LOOPS [cm] |
LOOP DIAMETER [cm] |
|---|---|---|
| 3 | 10-18 | 2 lub 4 |
| 4 | 16-20 | 2 lub 4 |
| 4.8 | 20-30 | 2 lub 4 |
| 5 | 24-30 | 2 lub 4 |
| 6 | 24-30 | 2 lub 4 |
| 7 | 24-30 | 2 lub 4 |
| 8 | 24-30 | 2 lub 4 |
| 8.5 | 24-30 | 2 lub 4 |
| 10 | 24-30 | 2 lub 4 |
3. INTENDED PURPOSE / INDICATIONS
The device is intended for short-term (up to 30 days) use to ensure the free flow of urine from the kidneys to the bladder in case of obstruction or narrowing of the ureter.
3.1. CONTRAINDICATIONS
The use of the device is usually contraindicated in the following cases:
- bladder fistula;
- bladder outlet obstruction;
- spastic / neurogenic bladder;
- coagulation disorders or the use of anticoagulants;
- allergy or hypersensitivity to polyurethane.
3.2. POTENTIAL ADVERSE EVENTS
Adverse events that may be associated with the use of the device include (in alphabetical order), but are not limited to:
- allergic reaction or hypersensitivity to polyurethane;
- bleeding;
- catheter dislocation;
- catheter fracture;
- catheter incrustation, malfunction and possible retention;
- fever;
- hematuria;
- infection / pyuria;
- pain;
- perforation of the renal pelvis / ureter;
- septic shock;
- symptoms of irritation of the bladder;
- uretero-arterial fistula;
- urinary incontinence / urgency;
- urine extravasation;
- vesico-renal reflux.
Any serious incident that has occurred in relation to the device should be reported to the manufacturer at reklamacje@balton.pl and the competent authority of the country in which the user and/or patient is established.
3.3. INTENDED USER PROFILE
Intended users of this device are only physicians who have received appropriate training in endourological procedures.
3.4. USE ENVIRONMENT
Use of this device is allowed only in healthcare facilities prepared for this kind of urological procedures.
3.5. PATIENT TARGET GROUP
Target group are patients with impaired free flow of urine from the kidneys to the bladder. No known data restricting use of this device in patients of particular gender or race. Before making decision concerning use of this device, potential benefits and risks should be considered individually for every patient.
4. WARNINGS
- This device is designed for single use in a single patient only. Do not resterilize or reuse. Reuse or resterilization may compromise the structural integrity of the device and/or lead to device failure which, in turn, may result in patient injury, illness or death. Reuse or resterilization may also create a risk of contamination of the product and cause patient infection including the transmission of infectious disease(s). Contamination of the product may lead to injury, illness or death of the patient.
- Do not use if the package is opened or damaged.
- Do not use after expiration date given on the package label.
- Do not use if labeling is incomplete or illegible.
6. HOW SUPPLIED
6.1. CONTENT OF THE PACKAGING
One (1) ureteral stenting kit in a blister / one (1) ureteral double pigtail catheter in a pouch, packed with Instructions for Use in a case box.
6.2. STERILITY
This product is supplied sterilized with ethylene oxide gas, in a blister / pouch. Only the content of the blister / pouch should be considered sterile. The device is only sterile if this packaging is not opened or damaged.
7. HANDLING AND STORAGE
Store at room temperature in a dry place, in the case box, as supplied. Do not expose to temperatures outside the range: 10 ºC ÷ 30 ºC.
8. DISPOSAL INSTRUCTIONS
Used product shall be treated as medical waste. After use, dispose the product and packaging in accordance with healthcare facilities, administrative and/or local government policy.
9. WARRANTY
If delivered product is damaged or has any other defects, inform the manufacturer and keep the device with original packaging.
10. OPERATIONAL INSTRUCTIONS
Balton sp. z o.o. shall not be liable for any direct, incidental or consequential damages resulting from the misuse of this product.
- Check the primary packaging (blister / pouch) for possible damage and expiry date.
- Open the package and take out the device.
- Check if the device is not damaged.
- Prepare the component, maintaining sterility.
- Disinfect the perineum according to procedures.
- Cover the patient with sterile drape.
- Remove the guidewire from the protector.
- Put the guidewire into the catheter, until both pigtails are straightened.
- Close the clamp immobilizing the catheter on the guidewire.
- Put the pusher onto the guidewire and advance until it contacts the catheter.
- Close the second clamp immobilizing the pusher on the guidewire.
- Insert the cystoscope into the bladder.
- Insert the prepared kit into the cystoscope, using driving ring.
- Drive the guidewire with the catheter through the ureter, into the renal pelvis.
- Open both clamps.
- Keeping the catheter in the renal pelvis with the use of the pusher, gently withdraw the guidewire and then the pusher.
- Confirm the correct position of the catheter in ultrasound, fluoroscopy or abdominal X-ray.
SYMBOLS GLOSSARY
-
CE mark
-
Medical device
-
Unique device identifier
-
Manufacturer
-
Date of manufacture
-
Use-by date
-
Catalogue number
-
Batch code
-
Consult instructions for use or consult electronic instructions for use
-
Do not use if the packaging is damaged and consult instructions for use
-
Caution
-
Sterilized using ethylene oxide
-
Do not resterilize
-
Do not re-use
-
Single sterile barrier system
-
Single sterile barrier system with protective packaging outside
-
Non-pyrogenic
-
Keep away from sunlight
-
Fragile, handle with care
-
Temperature limit
-
Not made with natural rubber latex
-
Keep dry
-
Recyclable packaging material
-
n units per package